Submitted by N.Y. Tan on Thu, 05/02/2015 - 00:17
Probing hydrogen bonding in liquids in the THz region with Debye and absorption analysis
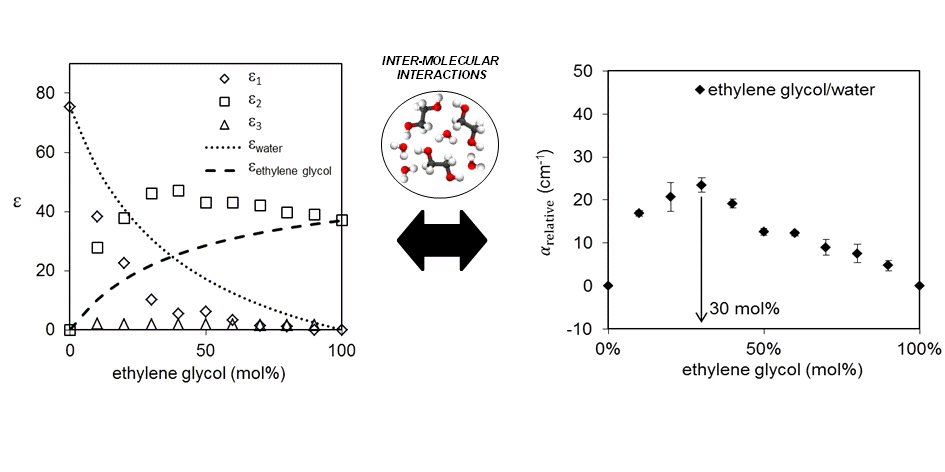
Our work on the THz spectra of hydrogen bonding liquids was recently published in Physical Chemistry Chemical Physics.
In this paper, we explored the hydrogen bonding characteristics of a wide variety of binary mixtures and analysed the results using direct absorption analysis and more complex Debye analysis. It was found when THz data are considered alone, Debye and absorption analysis provide the same qualitative information regarding hydrogen bonding interactions between the liquid components. Direct absorption analysis was applied systematically to a binary mixtures containing a variety of acetone derivatives and chloroform. The trends found matched well with theoretical predictions of hydrogen bond strength and affinity predicted based on the steric and electronic properties of the acetone derivatives.
Nicholas Y. Tan, Ruoyu Li, Pierre Bräuer, Carmine D'Agostino, Lynn F. Gladden and J. Axel Zeitler, Probing hydrogen-bonding in binary liquid mixtures with terahertz time-domain spectroscopy: a comparison of Debye and absorption analysis, Physical Chemistry Chemical Physics, doi: 10.1039/C4CP04477K, ASAP.
